medichem Meditrol FOB
Immunological stool rapid test
Item in stock
| Quantity | Price per unit | Price per unit (gross) | Reference price | Reference price (gross) |
|---|---|---|---|---|
| To 3 | €23.95 | €28.50 | €1.60 / 1 piece(s) | €1.90 / 1 piece(s) |
| From 4 | €22.95 | €27.31 | €1.53 / 1 piece(s) | €1.82 / 1 piece(s) |
| From 12 | €21.95 | €26.12 | €1.46 / 1 piece(s) | €1.74 / 1 piece(s) |
Meditrol FOB from medichem
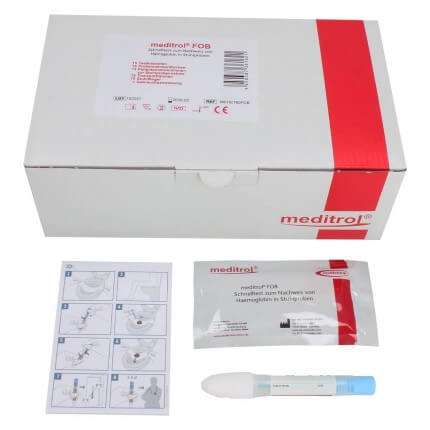
The meditrol FOB stool test from medichem is used for the qualitative detection of human hemoglobin in stool. The detection can be used as a tool for the diagnosis of colorectal cancer and enables early therapy. By means of visual color development on the test strip, hemoglobin present is clearly detected. The chromatographic rapid test is easy and user-friendly to use and the test result can be read after only 5 minutes.
Important note: For health professionals only.
Thetest may only be performed and interpreted by a physician or a person authorized by a physician. Medical laypersons may use and interpret the test incorrectly.
Product details
- Qualitative detection of human hemoglobin in stool
- Aids in the early detection of colorectal cancer
- Immunochromatographic, visual rapid test
- For professional in vitro diagnostic use
- High sensitivity and specificity
- Simple evaluation by interpretation of visual color development
- Test result available after only 5 minutes
- Fast and user-friendly application
- Integrated procedure control with control line
- Hygienic and easy to wipe off
- Storage temperature: 2 - 30 °C
Scope of delivery
- 1 package medichem meditrol FOB stool test à 15 tests consisting of:
- 15 test cassettes
- 15 sampling tubes
- 15 stool catchers
- 15 patient information for stool collection
- 15 transport bags
Return policy
This item is not eligible for returns.
For consumers, the right of withdrawal does not apply to contracts for the delivery of sealed goods that are not suitable for return for reasons of health protection or hygiene if their seal has been removed after delivery.
Brand: medichem
Further information

Manufacturer
medichem Vertriebs GmbH
Sandhof 8-10
24768 Rendsburg
Deutschland
info@medichem-online.de